Pharmaceutical traceability is the ability to track a medicine's real-time location in the supply chain (tracking) and reconstruct its complete history from manufacturing through to dispensing or patient use (tracing). It is governed by regulations including the US Drug Supply Chain Security Act (DSCSA), the EU Falsified Medicines Directive (FMD), and emerging frameworks across APAC, Latin America, and Africa — all built on GS1 serialization standards for interoperability.
Despite regulatory mandates, implementation maturity varies dramatically by region: the US has achieved full DSCSA serialization; over 90% of European pharmacies are connected to FMD verification systems; India has reached approximately 60% tracking coverage for specialty products; while much of Africa and Latin America faces significant infrastructure gaps. The WHO estimates that 10% of medicines in low- and middle-income countries are substandard or falsified, contributing to hundreds of thousands of deaths annually — making traceability not just a compliance exercise but a patient safety imperative. This guide maps the global landscape, explains the standards, and makes the case for treating traceability as a strategic platform rather than a regulatory checkbox.
Introduction
What traceability really means, where different markets stand today, and why it's becoming a strategic imperative
The discovery was shocking: what appeared to be legitimate cancer medications contained no active ingredients whatsoever. In 2012, counterfeit versions of Avastin—a life-saving cancer treatment—were distributed to medical practices across the United States, leaving patients to unknowingly receive ineffective treatments during critical treatment windows. The FDA ultimately sent warning letters to nearly 1,000 medical practices across 48 states as more counterfeit batches were uncovered.
This wasn't an isolated incident. The World Health Organization estimates that 10% of medicines in low- and middle-income countries are substandard or falsified, contributing to hundreds of thousands of deaths annually. The global counterfeit medicines market is worth an estimated $200-432 billion per year, making it one of the largest illicit markets worldwide.
Yet despite this staggering crisis, pharmaceutical traceability—the ability to track and trace medicines throughout the supply chain, remains poorly understood, inconsistently implemented, and often viewed through the narrow lens of regulatory compliance.
After years of implementing traceability solutions across multinational pharmaceutical companies, we've observed a fundamental disconnect: most executives think traceability is about scanning barcodes and meeting regulatory requirements. This narrow view is not only limiting the industry's response to counterfeiting but also missing massive opportunities for operational excellence and competitive advantage.
What is Pharmaceutical Traceability, Really?
Ask ten pharmaceutical executives to define traceability, and you'll likely get ten different answers. Some will mention barcodes, others will talk about serialization, and many will immediately jump to regulatory compliance. This confusion reveals how poorly the industry understands what traceability actually encompasses.
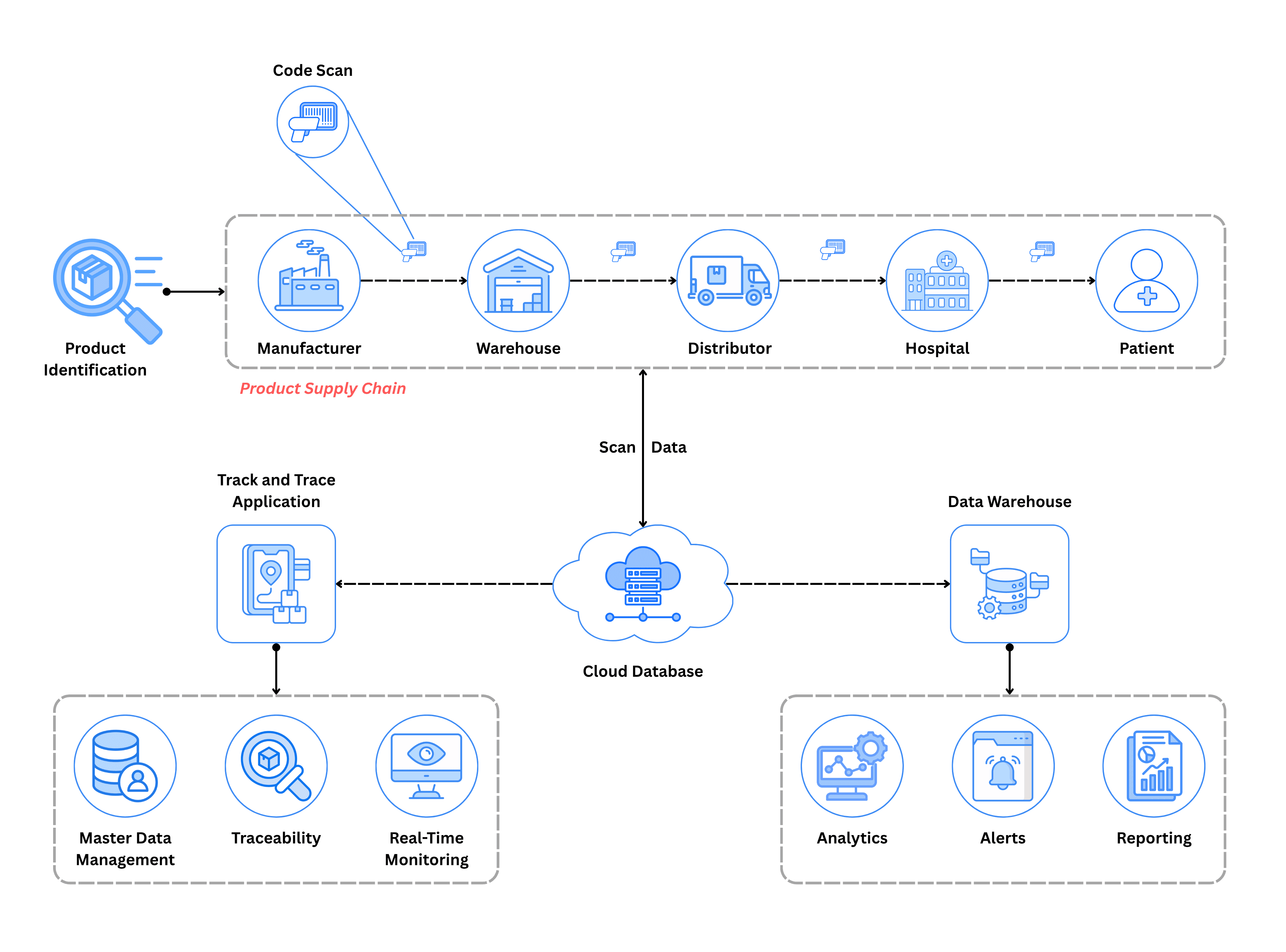
Traceability fundamentally consists of two components:
Tracking: Knowing where a product is right now—in which warehouse, at which pharmacy, with which patient.
Tracing: Understanding a product's complete history—where it's been, who handled it, how it moved through the supply chain.
But complete traceability goes far beyond these basic definitions. It's the foundation for an intelligent, connected pharmaceutical ecosystem that captures not just location data, but behavioral patterns, consumption trends, market dynamics, and operational insights that were previously invisible.
When traceability extends to the patient level, it intersects with therapy management — see how Zelthy's Complete Therapy Management (CTM) model unifies product tracking and patient assistance on a single platform.
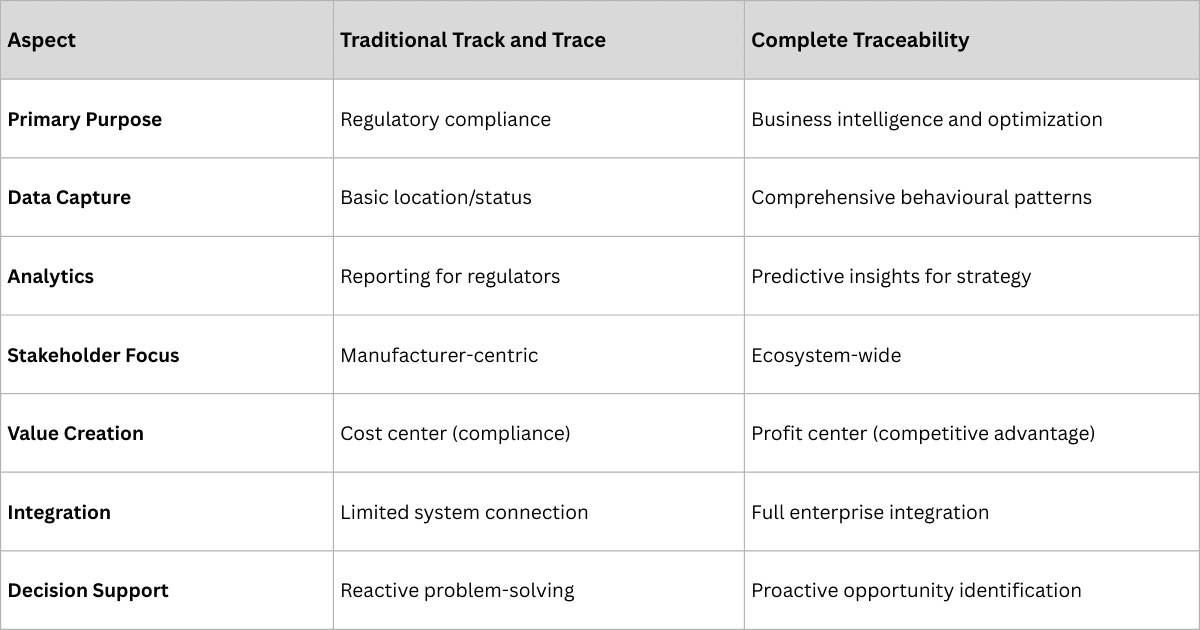
Traditional vs. Complete Traceability: Understanding the Difference
The Current Reality: A Fragmented Global Landscape
The implementation of pharmaceutical traceability varies dramatically across the globe, creating a patchwork of regulations, standards, and capabilities that reflects each region's unique challenges and priorities.
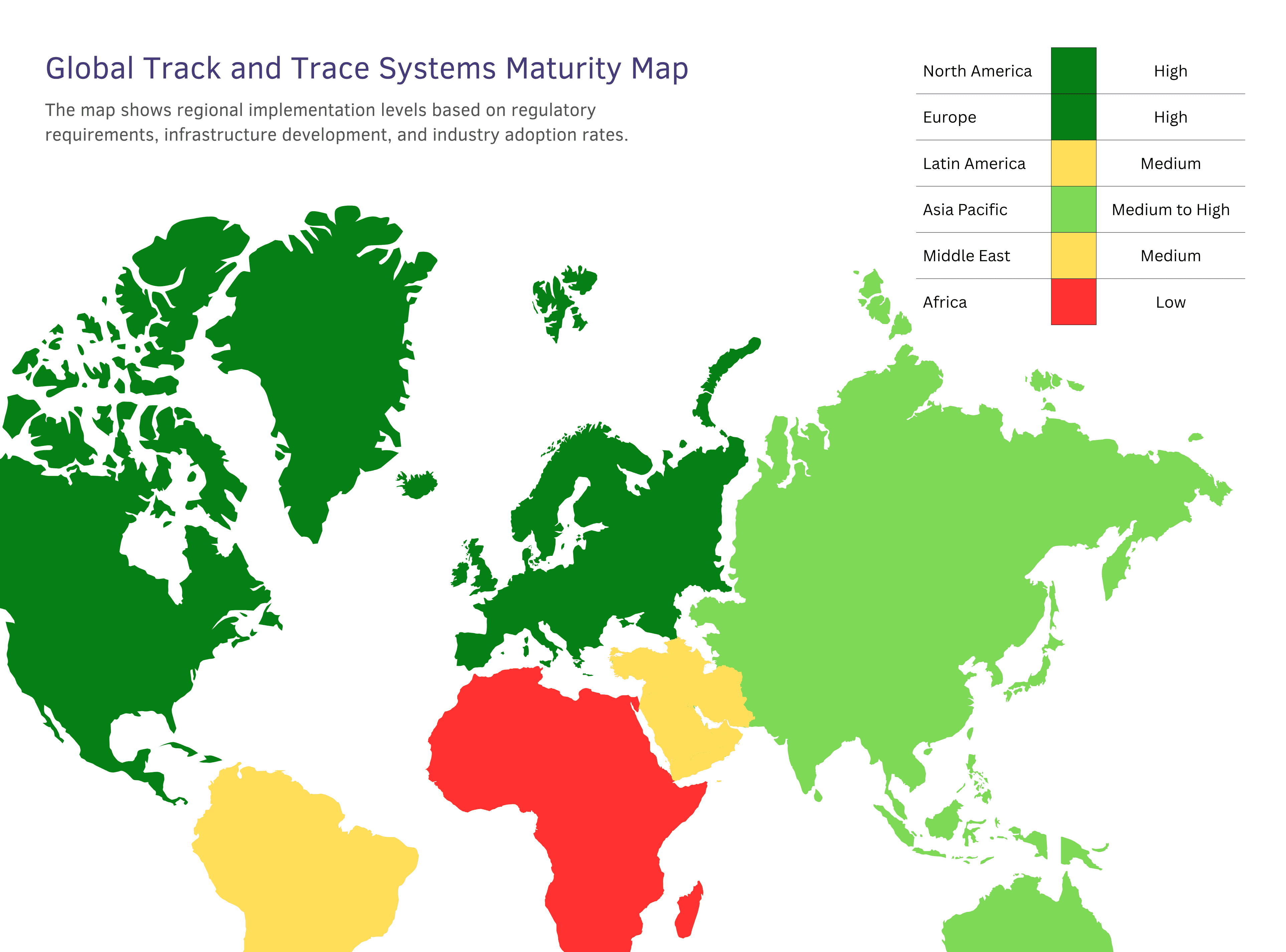
Global Track and Trace Systems Region-wise Maturity Levels
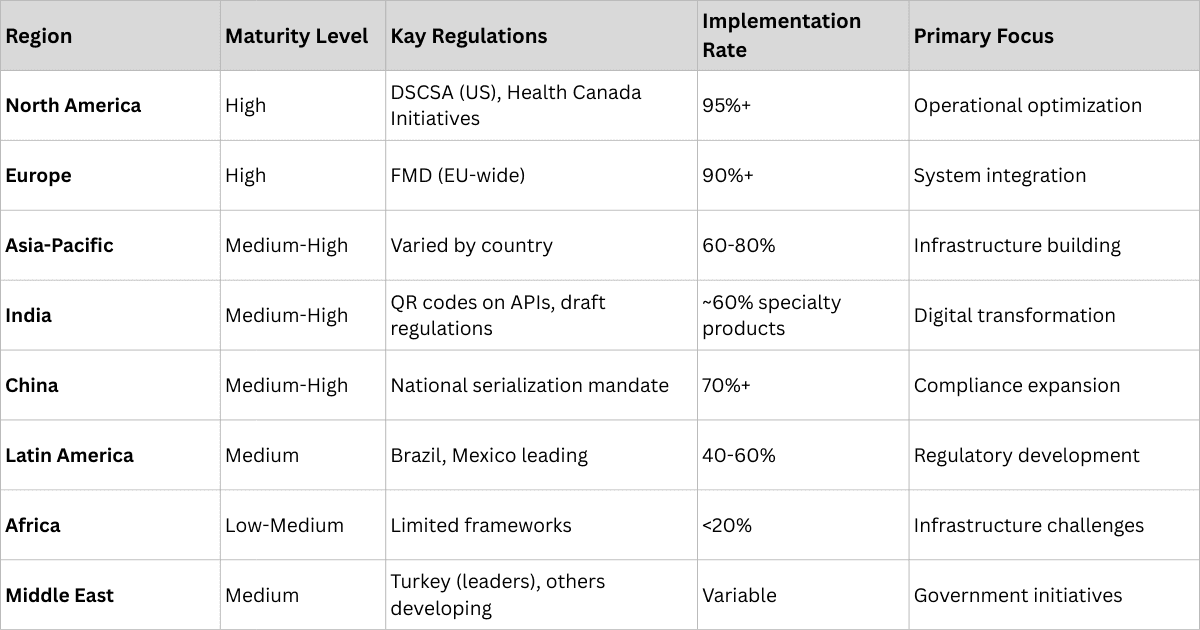
- High maturity markets (US, Europe) have achieved comprehensive regulatory compliance and focus on operational optimization.
- Medium maturity markets (India, China, Brazil) are rapidly implementing systems with strong government support.
- Emerging markets (Africa, Southeast Asia, Latin America) face infrastructure challenges but show increasing adoption urgency due to high counterfeiting rates.
North America: The DSCSA Success Story
The United States has emerged as a global leader in pharmaceutical traceability through the Drug Supply Chain Security Act (DSCSA), enacted in 2013 with full implementation achieved by November 2023. The DSCSA requires:
- Standardized product identifiers using GS1 standards
- Electronic data interchange throughout the supply chain
- Verification capabilities at every level
- Complete serialization for prescription medicines
This comprehensive approach has created one of the world's most robust pharmaceutical traceability systems, though adoption quality varies significantly among smaller distributors and pharmacies.
Europe: The FMD Framework
Europe's Falsified Medicines Directive (FMD), effective since 2017, mandates serialization and authenticity verification for prescription medicines across all EU member states. The European system emphasizes:
- Unique identifiers on every medicine package
- Anti-tampering devices to detect interference
- Centralized authentication systems
- Real-time verification at dispensing
The FMD has achieved impressive adoption rates, with over 90% of European pharmacies now connected to verification systems.
Asia-Pacific: Rapid Evolution
The Asia-Pacific region presents a complex landscape of varying maturity levels:
India is implementing QR codes on Active Pharmaceutical Ingredient (API) containers and has achieved approximately 60% tracking coverage for specialty products. The government's push for digital transformation has accelerated adoption, particularly among larger pharmaceutical companies.
China has implemented comprehensive serialization requirements for certain drug categories, with plans for expansion across all prescription medicines by 2025.
Japan maintains sophisticated traceability systems primarily focused on high-value and biologics products.
Emerging Markets: The Opportunity and Challenge
Latin America, Africa, and other emerging markets face the greatest counterfeiting challenges but also the most significant implementation obstacles:
Brazil and Mexico are advancing serialization requirements, though infrastructure limitations create implementation challenges.
African countries have limited traceability adoption due to infrastructure constraints, despite facing the highest rates of counterfeit medicines.
Russia operates the Chestny ZNAK system, requiring serialization for certain pharmaceutical products with plans for expansion.
Understanding the Standards: The GS1 Foundation
Global implementation of pharmaceutical traceability relies heavily on GS1 standards, developed by GS1—a global organization that creates standards for supply chain management and ensures interoperability across different systems and geographies.
Key GS1 Standards in Pharmaceuticals:
Global Trade Item Number (GTIN): Uniquely identifies products, enabling consistent tracking across all supply chain participants. GTINs are mandated by regulatory frameworks like the DSCSA.
Serial Shipping Container Code (SSCC): Identifies logistics units like pallets or cases, enabling efficient tracking of bulk shipments.
Global Location Number (GLN): Identifies specific locations such as manufacturing sites, warehouses, or pharmacies, ensuring precise tracking of product movement.
Electronic Product Code (EPC): Supports RFID-based data capture for real-time tracking and automated data collection.
These standards facilitate seamless data sharing across global supply chains. For example, a medicine manufactured in India using GTIN identification can be tracked through European distribution networks and verified at American pharmacies using the same standardized data structure.
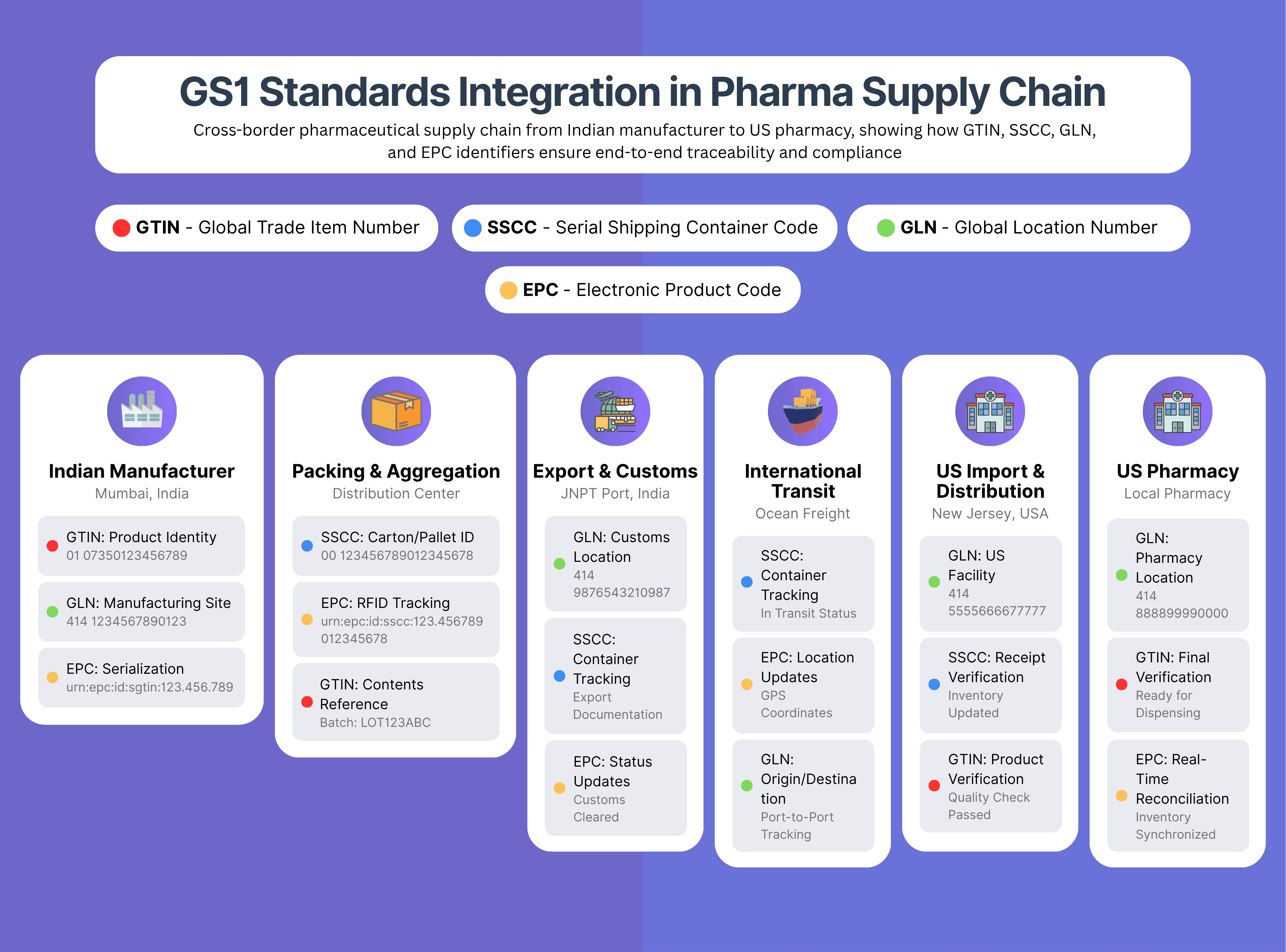
GS1 Standards Implementation by Market
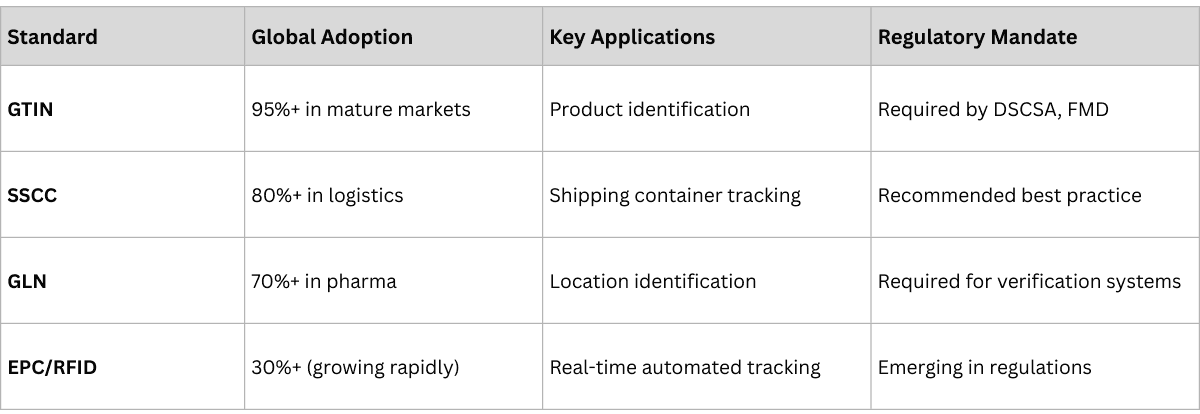
GS1 standards enable global interoperability: the US mandates GTIN and EPCIS under DSCSA, Europe requires DataMatrix codes under FMD, India implements QR codes under DCTS, Russia operates the Chestny ZNAK (MDLP) system, and Brazil is advancing SNCM serialization requirements.
Automatic Identification and Data Capture (AIDC) Technologies
GS1's AIDC technologies, particularly 2D DataMatrix codes, have become the backbone of modern pharmaceutical traceability. These codes can include:
- Product identification numbers
- Lot/batch numbers
- Expiration dates
- Serial numbers
This information enables automated data capture throughout the supply chain, reducing manual errors and ensuring comprehensive tracking capabilities.
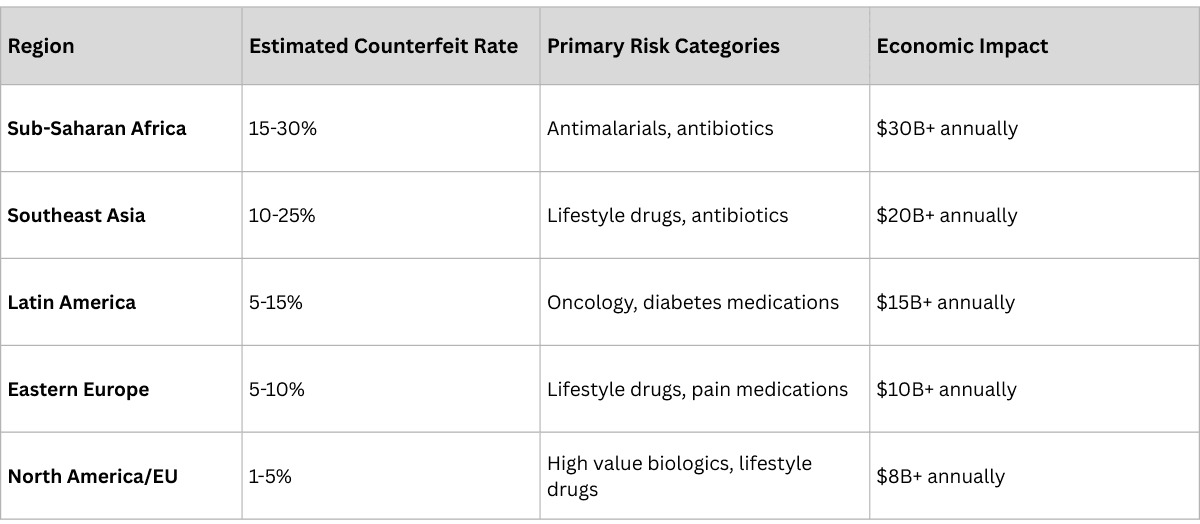
The Counterfeiting Crisis: Why Traceability is no longer Optional
The scale of pharmaceutical counterfeiting has reached crisis proportions, making traceability not just a regulatory requirement but a patient safety imperative.
By the Numbers:
- $200 billion: Estimated annual value of the global counterfeit medicines market
- 10%: Percentage of medicines that are substandard or falsified in low and middle-income countries
- 1 million: Estimated annual deaths caused by counterfeit medicines
- 200+: Countries affected by pharmaceutical counterfeiting
Real-World Impact Stories
In 2012, counterfeit Avastin (a cancer treatment) containing no active ingredients reached medical practices across the United States, putting cancer patients' lives at risk when they received ineffective treatments during critical treatment windows. The FDA ultimately identified nearly 1,000 affected medical practices across 48 states.
Recent incidents in Southeast Asia and Africa have involved counterfeit antimalarial medications containing incorrect active ingredients, leading to treatment failures and contributing to drug resistance in regions already struggling with malaria control.
These incidents highlight a sobering reality: counterfeiting isn't just an economic problem—it's a public health crisis that demands systematic solutions.
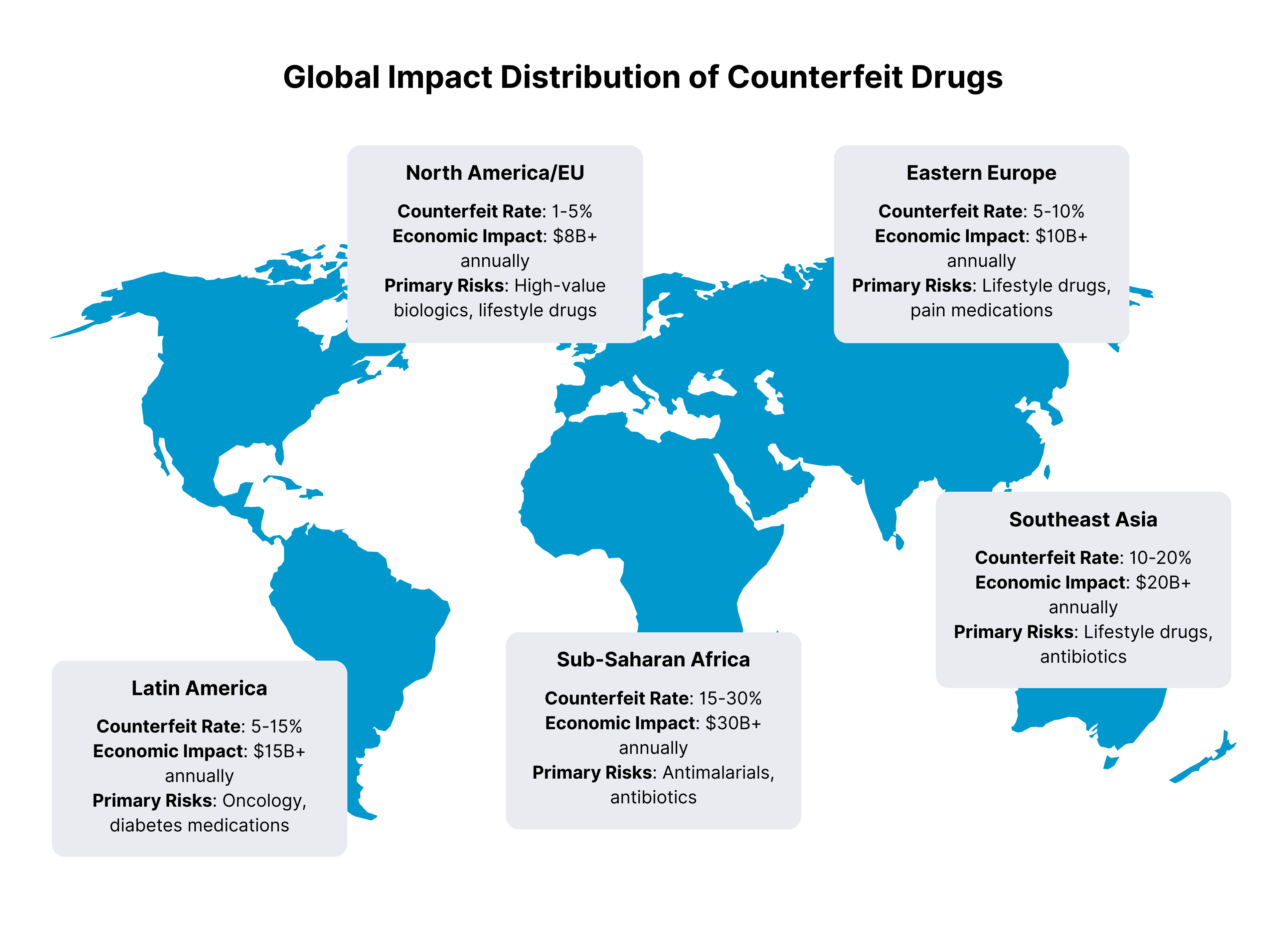
Impact of Counterfeiting by Region
Current Market Dynamics and Growth Projections
The pharmaceutical traceability market is experiencing unprecedented growth, driven by regulatory requirements, patient safety concerns, and increasing recognition of operational benefits.
Market Size and Projections:
- 2024: $5.19 billion global market size
- 2033: Projected $11.9 billion market size
- CAGR: 9.6% compound annual growth rate
Key Growth Drivers:
- Regulatory mandates expanding globally
- Increasing counterfeit incidents
- Digital transformation initiatives
- Supply chain optimization demands
- Patient safety awareness
Technology Adoption Trends:
- Cloud-based platforms gaining dominance
- AI and machine learning integration increasing
- Blockchain pilots expanding beyond proof-of-concept
- IoT integration for environmental monitoring
- Mobile applications becoming standard
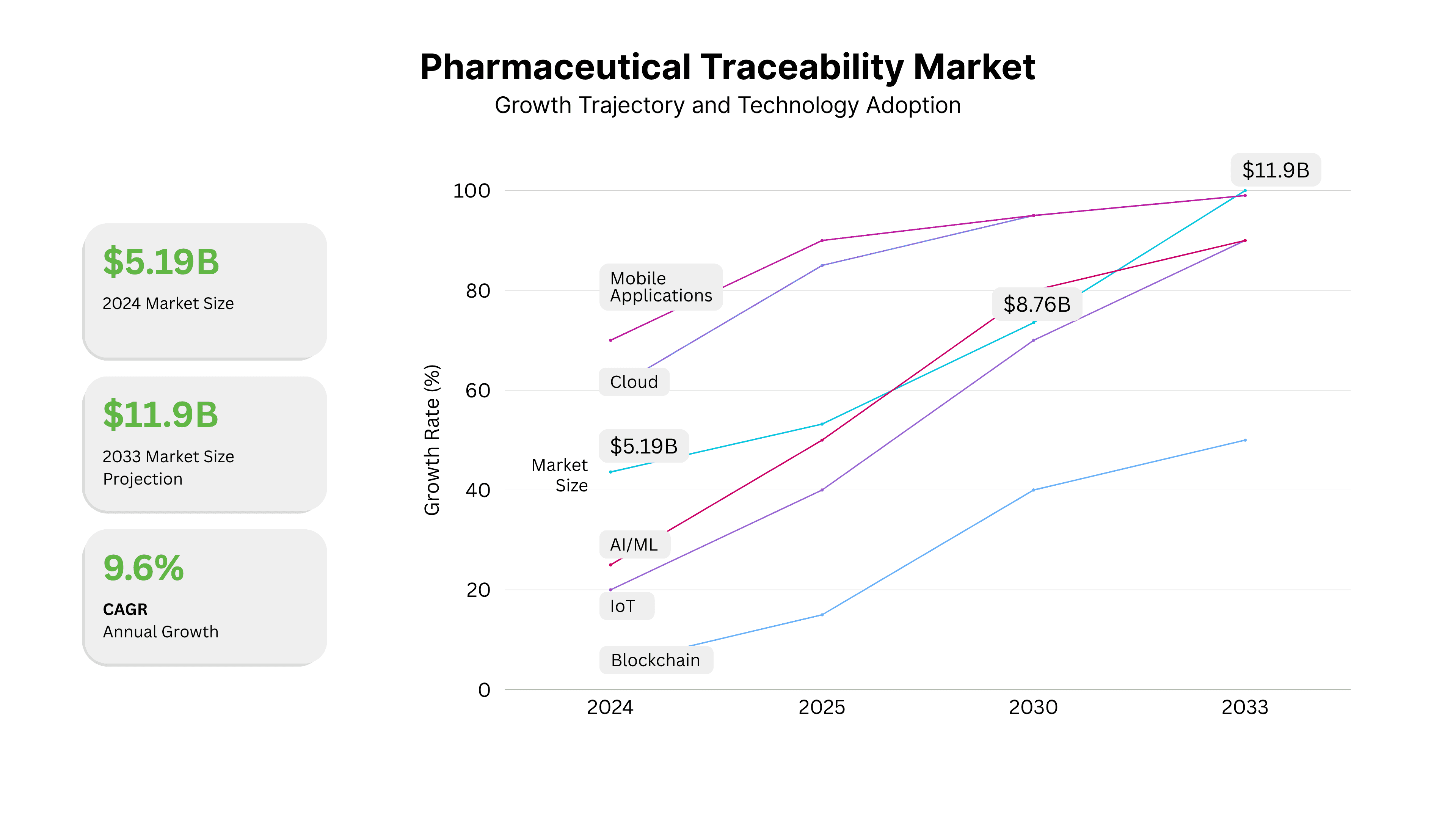
Technology Adoption Timeline
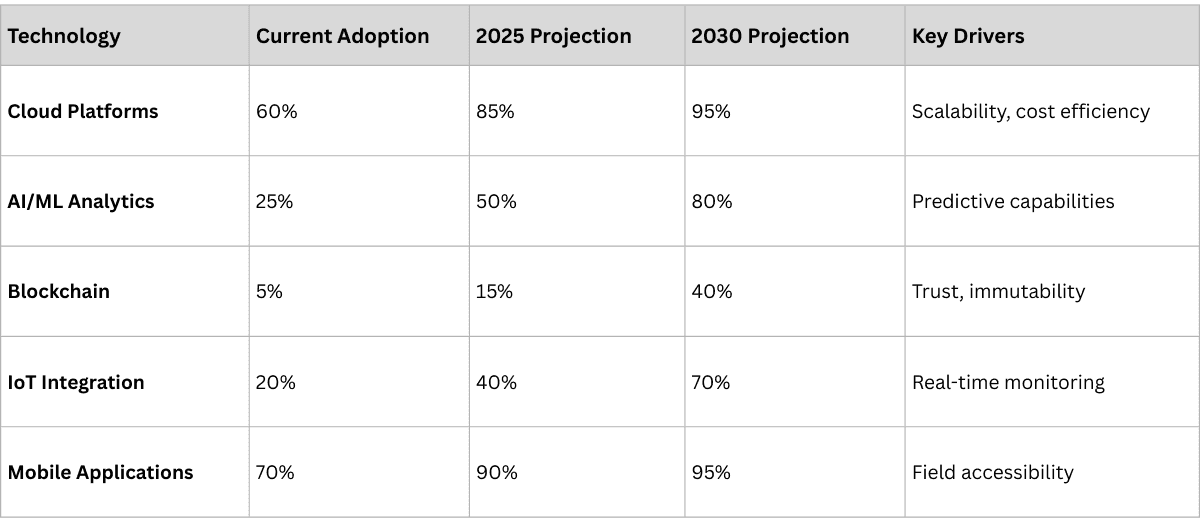
Regional Maturity Assessment: Where We Stand Today
Our implementation experience across multiple markets reveals significant variations in traceability maturity:
High Maturity Markets
United States and Europe have achieved comprehensive regulatory compliance and are now focusing on operational optimization and competitive advantage applications. These markets feature:
- Universal serialization implementation
- Advanced analytics adoption
- Partner ecosystem integration
- Service-based business model exploration
Medium Maturity Markets
India, China, and Brazil are rapidly implementing comprehensive systems with strong government support. These markets show:
- Accelerating adoption rates
- Infrastructure modernization
- Significant transformation opportunities
- Growing private sector investment
Emerging Maturity Markets
Many African, Southeast Asian, and smaller Latin American countries face infrastructure challenges but demonstrate increasing awareness and government interest. Key characteristics include:
- Limited current adoption
- High counterfeiting rates creating urgency
- Leapfrog technology opportunities
- International aid and partnership initiatives
The Implementation Reality: Common Challenges and Success Factors
Despite clear benefits and regulatory requirements, pharmaceutical traceability implementation faces consistent challenges across all markets:
Universal Challenges
- Stakeholder Alignment: Coordinating manufacturers, distributors, pharmacies, and regulators
- Technology Integration: Connecting new traceability systems with existing ERP, MES, and other platforms
- Data Management: Handling massive volumes of real-time data while ensuring security and privacy
- Cost Considerations: Balancing implementation costs with long-term benefits
- Change Management: Training personnel and adapting operational processes
Success Factors from Leading Implementations
- Early Stakeholder Engagement: Including all supply chain participants in planning phases
- Phased Implementation: Starting with high-value products before full-scale deployment
- User-Friendly Technology: Ensuring systems are intuitive for field personnel
- Comprehensive Training: Providing ongoing education and support
- Clear Value Proposition: Demonstrating benefits beyond compliance
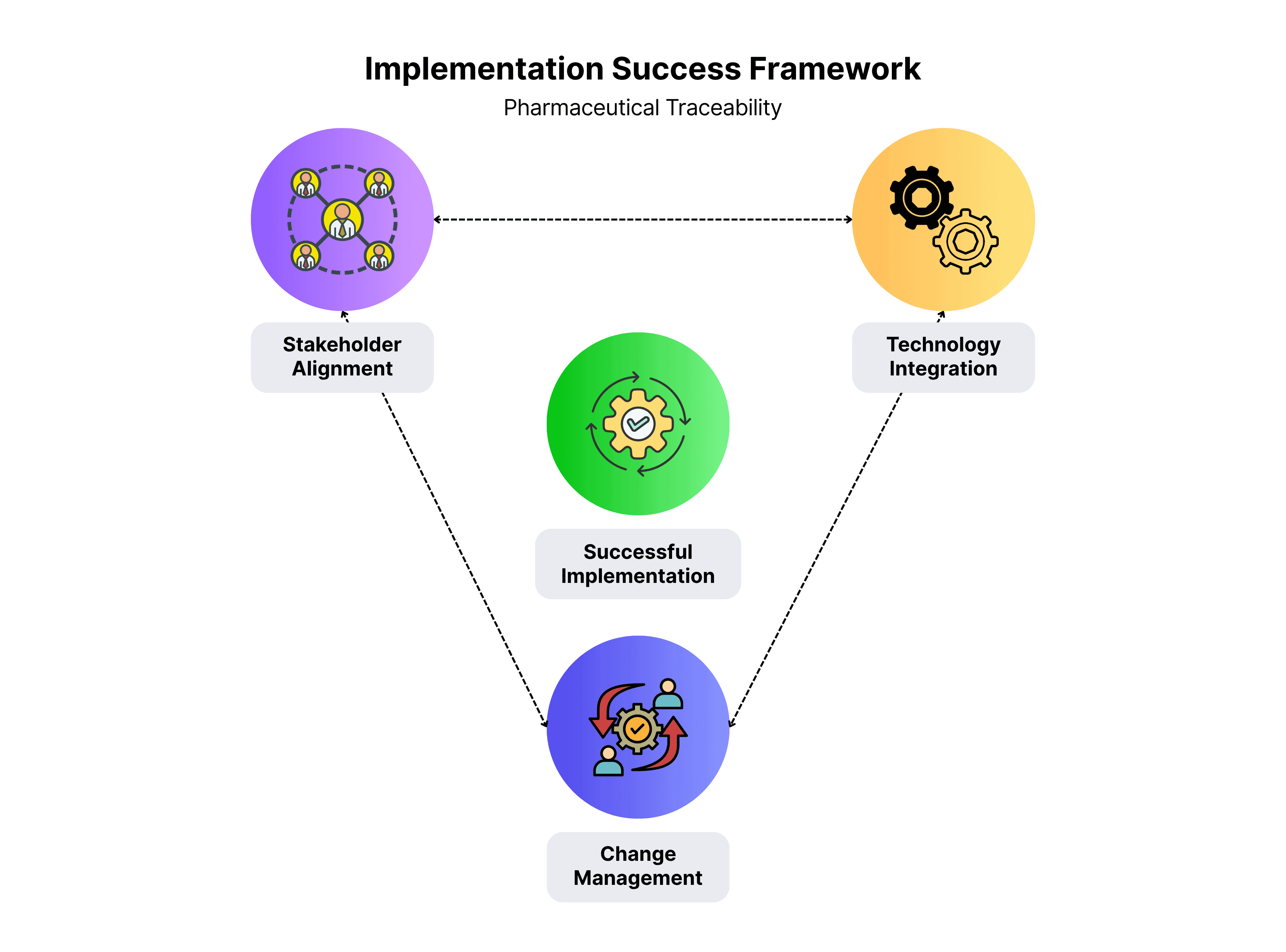
Implementation Challenges vs. Success Factors
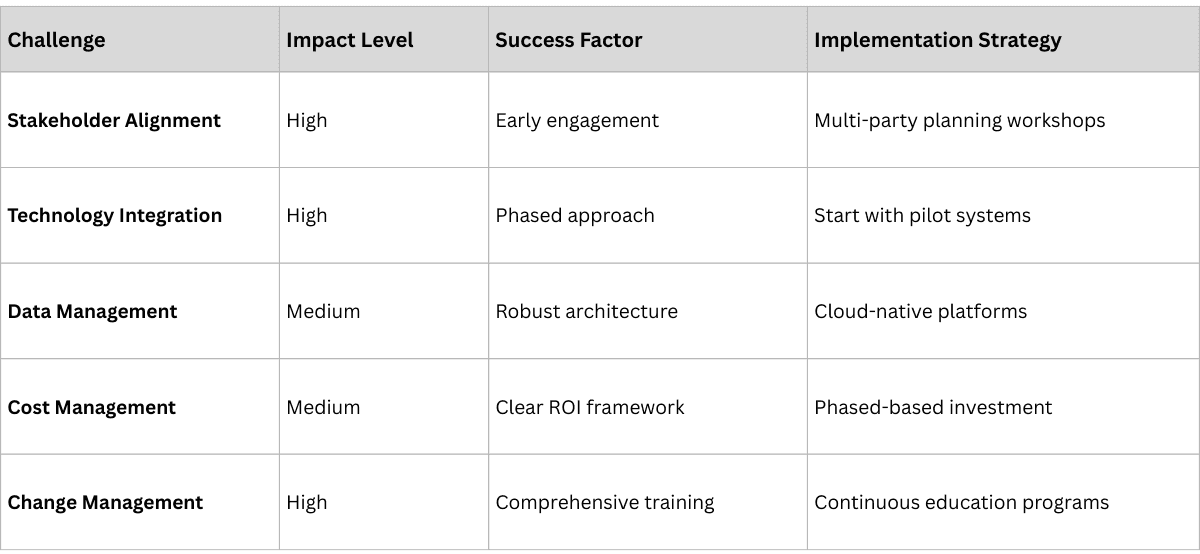
Universal implementation challenges include:
- stakeholder alignment across manufacturers, distributors, and pharmacies
- technology integration with legacy ERP and MES systems
- managing massive real-time data volumes
- and balancing implementation costs with long-term benefits.
Successful implementations counter these through:
- early stakeholder engagement
- phased rollouts starting with high-value products
- user-friendly technology
- comprehensive training
- and clear value propositions beyond compliance.
Technology Trends Shaping the Future
Several emerging technologies are enhancing pharmaceutical traceability capabilities and expanding potential applications:
Artificial Intelligence and Machine Learning: AI is enabling predictive analytics for demand forecasting, pattern recognition for fraud detection, and automated decision-making for supply chain optimization. Early adopters report significant improvements in demand prediction accuracy and inventory management.
Blockchain Integration: While still largely in pilot phases, blockchain promises immutable audit trails, enhanced trust frameworks for partner collaboration, and smart contracts for automated transactions. Several pharmaceutical companies are exploring blockchain for critical product authentication.
Internet of Things (IoT): IoT sensors enable environmental monitoring throughout the supply chain, real-time condition tracking for temperature-sensitive products, and automated data capture that reduces manual processes. This is particularly valuable for biologics and other sensitive medications.
Advanced Analytics Platforms: Modern analytics platforms can process massive data streams in real-time, provide geographic and demographic segmentation, enable competitive intelligence gathering, and optimize ROI across all business functions.
Looking Ahead: The Next Decade of Pharmaceutical Traceability
Based on current trends and our implementation experience, several developments will shape pharmaceutical traceability over the next decade:
Regulatory Evolution: Expect expanding mandates in emerging markets, harmonization of global standards, and increased focus on patient-level tracking.
Technology Maturation: AI and blockchain will move from pilots to production, IoT integration will become standard, and platforms will become increasingly sophisticated.
Business Model Innovation: Companies will increasingly view traceability as a platform for competitive advantage rather than just compliance, new revenue streams will emerge from traceability capabilities, and service-based models will expand.
Global Standardization: International cooperation will increase, technology platforms will become more interoperable, and best practices will spread more rapidly across markets.
The Strategic Imperative
The global state of pharmaceutical traceability reveals an industry in transition. While regulatory compliance has driven initial adoption, leading companies are discovering that traceability offers far more than regulatory checkbox completion.
The pharmaceutical companies that recognize traceability as a strategic capability—not just a compliance requirement—will be better positioned to compete in an increasingly complex global market. They'll have superior operational intelligence, deeper customer relationships, and new revenue opportunities that competitors struggle to replicate.
As the global pharmaceutical industry continues to evolve, traceability will increasingly separate market leaders from followers. The question isn't whether to implement traceability—regulatory requirements have made that decision. The question is whether to implement it strategically or merely tactically.
The foundation is being laid today for the pharmaceutical supply chains of tomorrow. Companies that understand this reality and act accordingly will shape the future of pharmaceutical traceability—and capture the competitive advantages that come with it.
Companies ready to move from compliance-first traceability to operational intelligence can explore Zelthy's end-to-end pharma traceability platform — India's largest track-and-trace provider for life sciences, with live deployments across 12+ countries.
Next in the series: The Executive's Implementation Guide to Complete Traceability — the proven strategies and common pitfalls that determine whether traceability projects deliver ROI or become expensive compliance exercises.
About This Series
This blog post is part of "The Complete Guide to Pharmaceutical Traceability" series, drawing from our experience implementing traceability solutions across multinational pharmaceutical companies in India, Europe, and North America. Each post builds on real-world insights from deployments serving millions of patients and processing billions of pharmaceutical transactions.
Upcoming in This Series:
- Blog 2: The Executive's Implementation Guide to Complete Traceability
- Blog 3: How Traceability Transforms Pharma Business Models
- White Paper: Beyond Track and Trace - Strategic Business Model Transformation
For specific questions about traceability requirements in your market or to discuss implementation strategies, feel free to reach out to our team at connect@zelthy.com or send us a DM on LinkedIn.
Frequently Asked Questions
What is pharmaceutical traceability?
Pharmaceutical traceability is the ability to track and trace medicines throughout the entire supply chain — from manufacturing through distribution to the patient — by capturing location, custody, and handling data at every step. It consists of two components: tracking (knowing where a product is right now) and tracing (reconstructing a product's complete custody history). Complete traceability extends beyond these basics to generate intelligence on consumption patterns, market dynamics, and patient behavior.
How large is the counterfeit medicines problem globally?
The WHO estimates that 10% of medicines in low- and middle-income countries are substandard or falsified, contributing to an estimated one million deaths annually. The global counterfeit medicines market is valued at $200–432 billion per year, making it one of the world's largest illicit markets. High-profile incidents — such as counterfeit Avastin cancer medication containing no active ingredients reaching nearly 1,000 US medical practices in 2012 — illustrate the direct patient safety consequences of traceability gaps.
What are GS1 standards and why are they foundational to pharma traceability?
GS1 is a global organization that develops supply chain identification standards used across pharmaceutical traceability systems worldwide. Key standards include the Global Trade Item Number (GTIN) for product identification, the Serial Shipping Container Code (SSCC) for logistics units, and the Global Location Number (GLN) for supply chain locations. GS1 standards are mandated by both DSCSA and FMD, ensuring that serialized product data is interoperable across manufacturers, distributors, pharmacies, and regulators in different countries.
Where does Asia-Pacific stand on pharmaceutical traceability maturity?
Asia-Pacific presents varied maturity levels. India has implemented QR codes on API containers and achieved approximately 60% tracking coverage for specialty products. China has serialization requirements for certain drug categories with planned expansion across all prescription medicines. Japan maintains sophisticated systems for high-value biologics. Emerging markets in Southeast Asia face significant implementation gaps despite high counterfeit exposure — infrastructure constraints, not regulatory intent, are the primary adoption barrier.
What is the pharmaceutical traceability market size and growth rate?
The global pharmaceutical traceability market was valued at $5.19 billion in 2024 and is projected to reach $11.9 billion by 2033, representing a 9.6% compound annual growth rate. Growth is driven by expanding global regulatory mandates, increasing counterfeit incidents, and growing recognition that traceability infrastructure delivers operational and commercial value beyond compliance. Cloud-based platforms, AI-driven analytics, and IoT integration for environmental monitoring are the leading technology adoption trends.
What is the difference between serialization and aggregation in pharma traceability?
Serialization assigns a unique identifier to each individual medicine unit — the minimum requirement under DSCSA and FMD. Aggregation maps those unit-level identifiers to their parent cases and pallets, creating a hierarchical relationship across the supply chain. Aggregation is not mandated by most regulations, but it is the data layer that enables supply chain analytics, demand sensing, diversion detection, and recall efficiency. Without aggregation, serialization achieves compliance but not operational intelligence.
How does pharmaceutical traceability differ from track-and-trace in other industries?
Unlike retail or logistics track-and-trace — which primarily optimizes inventory and delivery — pharmaceutical traceability carries direct patient safety and regulatory obligations. Every serialized medicine must be verifiable at the point of dispensing to detect counterfeits and prevent diversion. Adverse event reporting, cold-chain compliance, and recall management create additional data requirements absent in other industries. Regulatory mandates (DSCSA, FMD) also impose specific data standards, retention requirements, and interoperability obligations that exceed typical supply chain visibility programs.
References and Sources
- World Health Organization. "Substandard and Falsified Medical Products." WHO Fact Sheet, November 2022.
- United Nations Office on Drugs and Crime. "Trafficking in Medical Products in the Sahel." 2022.
- U.S. Food and Drug Administration. "Drug Supply Chain Security Act (DSCSA) Implementation." FDA Guidelines, Updated 2023.
- European Medicines Agency. "Falsified Medicines Directive - Implementation Guidelines." EMA Report, 2022.
- IMARC Group. "Track and Trace Solutions Market: Global Industry Trends, Share, Size, Growth, Opportunity and Forecast 2024-2033." Market Research Report, 2024.
- GS1 Global. "GS1 Healthcare Implementation Guidelines." Standards Documentation, 2023.
- National Library of Medicine. “Fighting the global counterfeit medicines challenge: A consumer-facing communication strategy in the US is an imperative.” 2022.
- Strategy&, PwC. “Fighting counterfeit pharmaceuticals: New defenses for an underestimated - and growing - menace.” 2017.
- Pharmaceutical Security Institute. "Counterfeit Incident Trends Report 2023." PSI Annual Report, 2024.
- Altunkan, Serap & Yasemin, Alper & Aykac, Ismail & Akpinar, Elgin. (2012). Turkish pharmaceuticals track & trace system. 24-30. DOI: 10.1109/HIBIT.2012.6209037
- Indian Ministry of Health and Family Welfare. "Central Drugs Standard Control Organization Guidelines for QR Coding." Government Guidelines, 2023.
- USAID. “Implementation Guidance for Pharmaceutical Traceability Leveraging GS1 Global Standards.” 2019.
- Prophecy Market Insights. "Global Pharmaceutical Anti-Counterfeiting Technologies Market Overview." 2024.
- World Health Organizations Atlas of African health statistics. Universal health coverage and the sustainable development goals in the WHO african region. 2018.
- Pharmaceutical Commerce. “DSCSA Compliance: The Time is Now." 2025.
- National Library of Medicine. “Tackling Counterfeit Drugs: The Challenges and Possibilities." 2023.
- International Bar Association. “Counterfeit Pharmaceuticals: Innovative Strategies for Combatting Global Health Threats." 2025.
- National Library of Medicine. “Falsified and substandard medicines trafficking: A wakeup call for the African continent." 2022.