Implementing pharmaceutical traceability requires a phased approach that treats the project as an organizational transformation, not a technology deployment. Successful implementations follow a five-phase framework: strategic foundation and stakeholder alignment (months 1–2), platform architecture and design (months 2–4), stakeholder enablement and change management (months 3–6), phased rollout (months 4–9), and continuous optimization (ongoing). Companies that follow this structured approach achieve compliance and operational transformation; those that treat traceability as a pure IT project consistently face adoption failures, timeline delays, and suboptimal results.
The failure patterns are well-documented: treating traceability as technology-only rather than organizational change, misalignment between manufacturers, distributors, and pharmacies, building for minimum compliance rather than strategic value, and underestimating change management. In one 11-site study, serialization capital costs averaged €600,000 per line — four times regulatory estimates — yet 88% of sites reported no net productivity gain. This guide provides the frameworks, phase-by-phase playbook, and hard-learned lessons from implementations across multinational pharmaceutical companies.
Introduction
Proven strategies, common pitfalls, and the roadmap that successful pharmaceutical companies follow
The pharmaceutical executive's frustration was palpable. "We've been working on this traceability project for eighteen months," she said. "We're millions over budget, six months behind schedule, and we still can't track a single product end-to-end. Where did we go wrong?"
This conversation, unfortunately, represents the reality many pharmaceutical companies face during complex technology implementations. While specific failure rates for traceability projects aren't definitively documented, industry experience shows that successful implementations require significantly more organizational transformation focus than technology focus. Companies that treat traceability as purely a technology project consistently struggle with adoption, timeline delays, and suboptimal results. Yet the 30% that succeed don't just meet compliance requirements—they transform their operations and create lasting competitive advantages.
After implementing complete traceability solutions across numerous multinational pharmaceutical companies, we've identified the critical factors that separate successful implementations from expensive failures. The difference isn't in the technology—it's in the approach, the planning, and the understanding that traceability implementation is fundamentally an organizational transformation, not just a technology deployment.
This guide provides pharmaceutical executives with the proven strategies, practical frameworks, and hard-learned lessons that ensure traceability implementations succeed on time, on budget, and with transformational results.
Why Most Traceability Implementations Fail
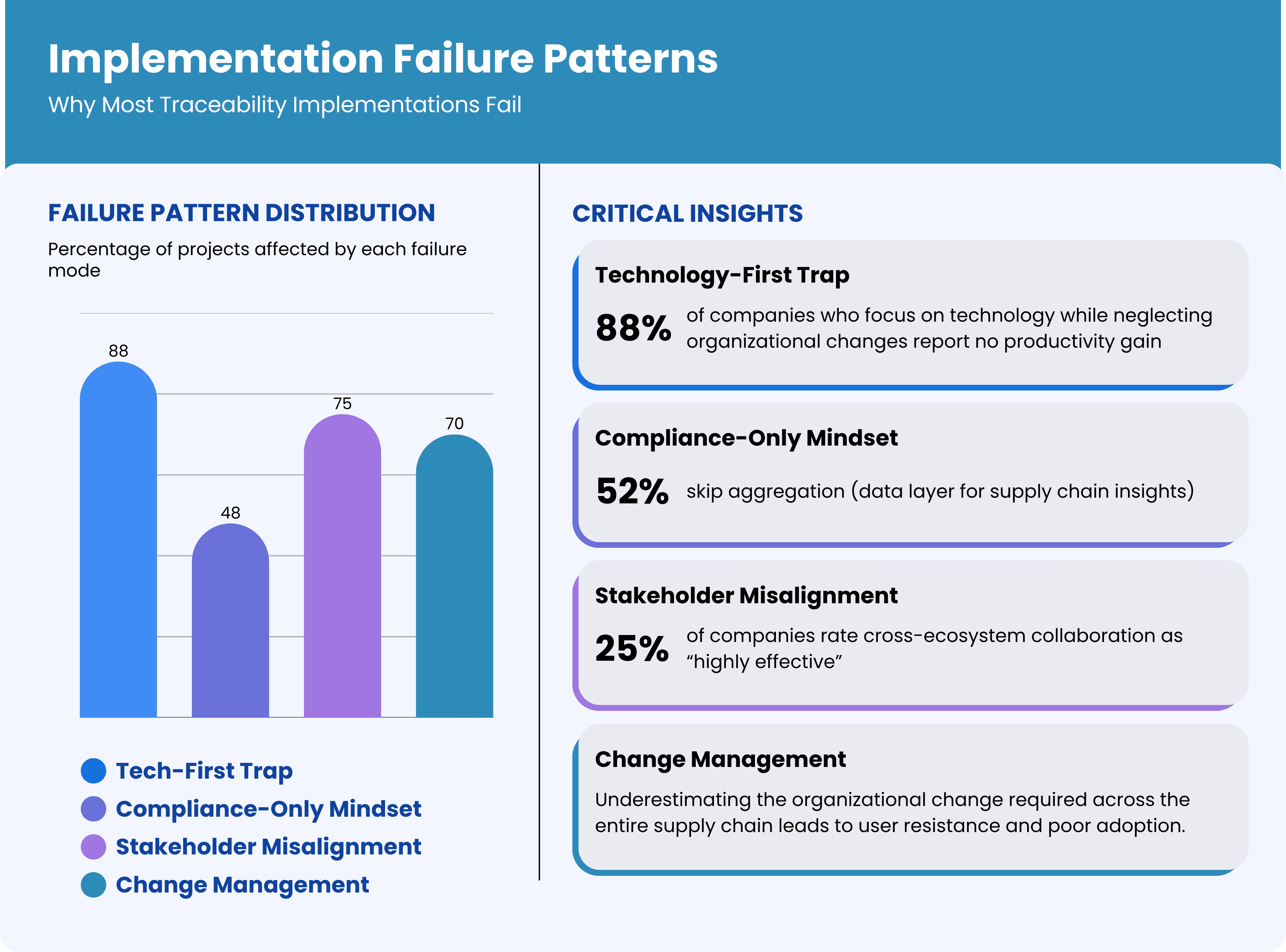
Before diving into success strategies, it's crucial to understand why so many traceability projects stumble. Our analysis of failed implementations reveals consistent patterns:
1. The Technology-First Trap
The most common failure mode is treating traceability as a pure technology implementation. Companies focus on selecting software platforms, installing hardware, and integrating systems while neglecting the organizational and process changes required for success. Technology is the enabler, not the solution. In one 11-site Irish study, serialization capital costs averaged €600,000 per line, four times regulatory estimates, yet 88% of sites reported no net productivity gain.
2. Stakeholder Misalignment
Pharmaceutical traceability involves multiple stakeholders—manufacturers, distributors, pharmacies, regulators—each with different priorities, systems, and incentives. Projects that don't address stakeholder alignment from day one inevitably face resistance, adoption problems, and integration challenges. End-to-end pharmaceutical traceability touches manufacturers, CMOs, 3PLs, wholesalers, hospitals, pharmacies, and regulators. Bain’s Global State of Traceability survey found only 25% of companies rate their cross-ecosystem collaboration as “highly effective”.
3. Compliance-Only Mindset
Companies that implement traceability solely for regulatory compliance build systems that meet minimum requirements but miss opportunities for operational improvement and competitive advantage. These implementations often become cost centers rather than value creators. A 2021 PMMI survey notes that 52% of pharma plants implementing US DSCSA standards skipped aggregation—the data layer required for actionable supply-chain insights—because “it wasn’t mandated”.
4. Underestimating Change Management
Traceability implementation requires significant changes to established workflows, processes, and behaviors across the entire supply chain. Companies that underestimate the change management component face user resistance, poor adoption, and suboptimal results.
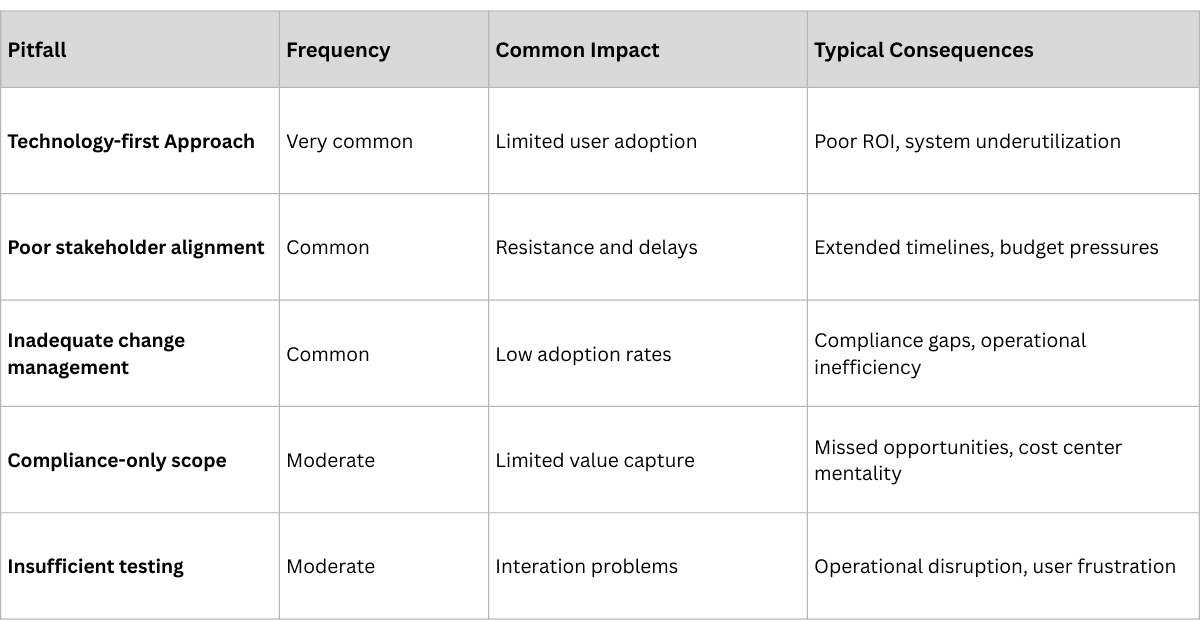
Common Implementation Pitfalls
The Proven Implementation Framework
Successful traceability implementations follow a structured approach that addresses technology, people, processes, and organizational alignment simultaneously. Our framework consists of five phases, each with specific objectives, deliverables, and success criteria.
Phase 1: Strategic Foundation (Months 1-2)
The foundation phase establishes the strategic context, defines success criteria, and aligns stakeholders around common objectives.
Key Activities:
- Current State Assessment: Comprehensive evaluation of existing tracking capabilities, system integrations, and process maturity
- Stakeholder Mapping: Identification of all internal and external stakeholders, their requirements, and success criteria
- Value Case Development: Quantification of business benefits beyond compliance, including operational improvements and competitive advantages
- Success Metrics Definition: Clear, measurable KPIs that define project success and ongoing value realization
Critical Decisions:
- Implementation scope and phasing strategy
- Technology platform selection criteria
- Organizational structure and governance model
- Budget allocation and resource commitment
Phase 1 Success Checklist:
- Complete stakeholder buy-in on project scope and objectives
- Quantified business case with clear ROI projections
- Defined success metrics and measurement framework
- Approved budget and resource allocation
- Project governance structure established
Success Indicator: All key stakeholders can articulate the project's strategic value beyond compliance
Phase 2: Platform Architecture and Design (Months 2-4)
The architecture phase focuses on technology platform selection, system design, and integration planning.
Technology Platform Selection Framework
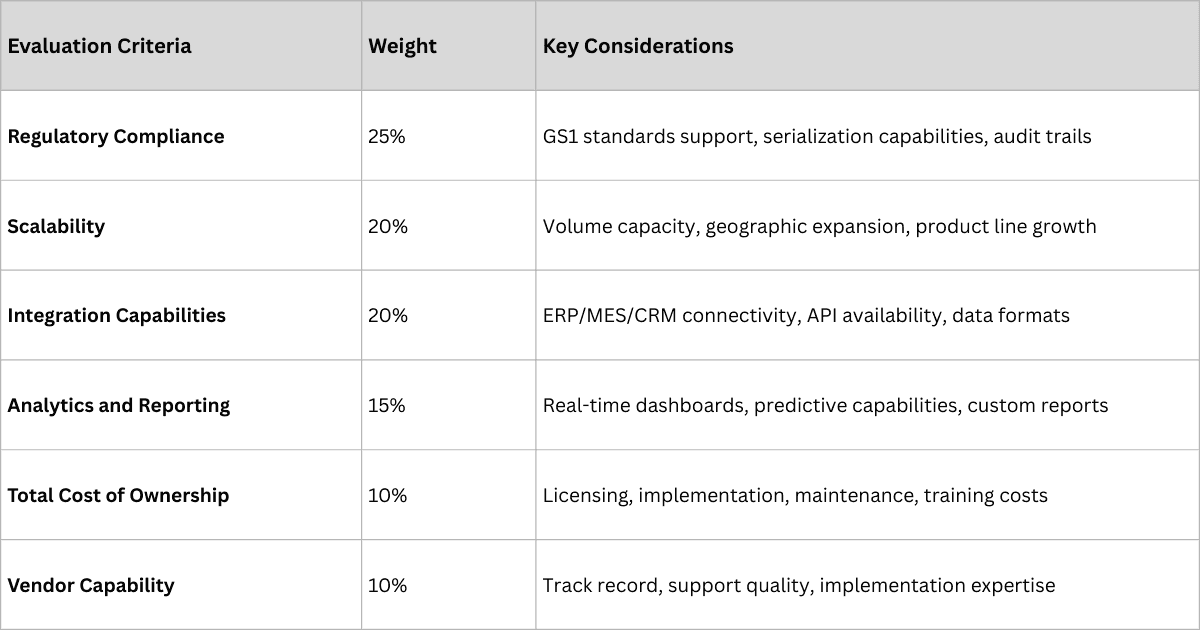
Architecture Design Principles
- Cloud-First Approach: Leverages scalability, security, and cost efficiency of modern cloud platforms
- API-Enabled Integration: Ensures seamless connectivity with existing and future systems
- Data Standardization: Implements GS1 standards for interoperability and compliance
- Security by Design: Embeds robust security controls throughout the platform architecture
Integration Planning:
Successful implementations require careful integration with existing enterprise systems:
Traceability Platform Integration Map:
ERP Systems → Product Master Data, Inventory Management
MES Systems → Production Data, Batch Records
CRM Systems → Customer Information, Sales Data
WMS Systems → Warehouse Operations, Shipping
Analytics → Business Intelligence, Reporting
Phase 2 Deliverables:
- Detailed technical architecture documentation
- Selected technology platform and vendor contracts
- Integration specifications and development plans
- Security and compliance framework
- Proof-of-concept validation results
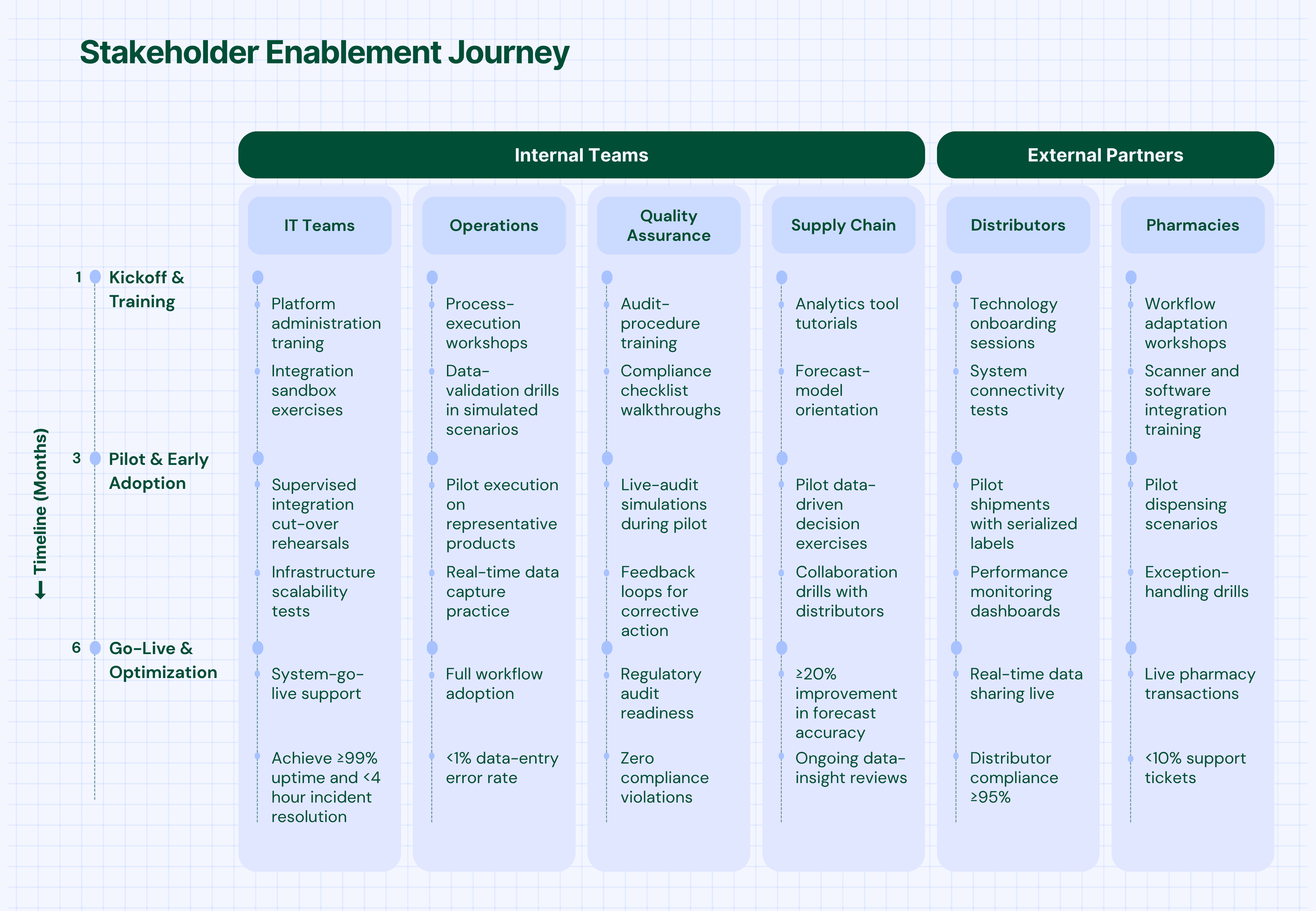
Phase 3: Stakeholder Enablement (Months 3-6)
The enablement phase focuses on preparing all stakeholders—internal teams and external partners—for successful adoption.
Internal Team Preparation
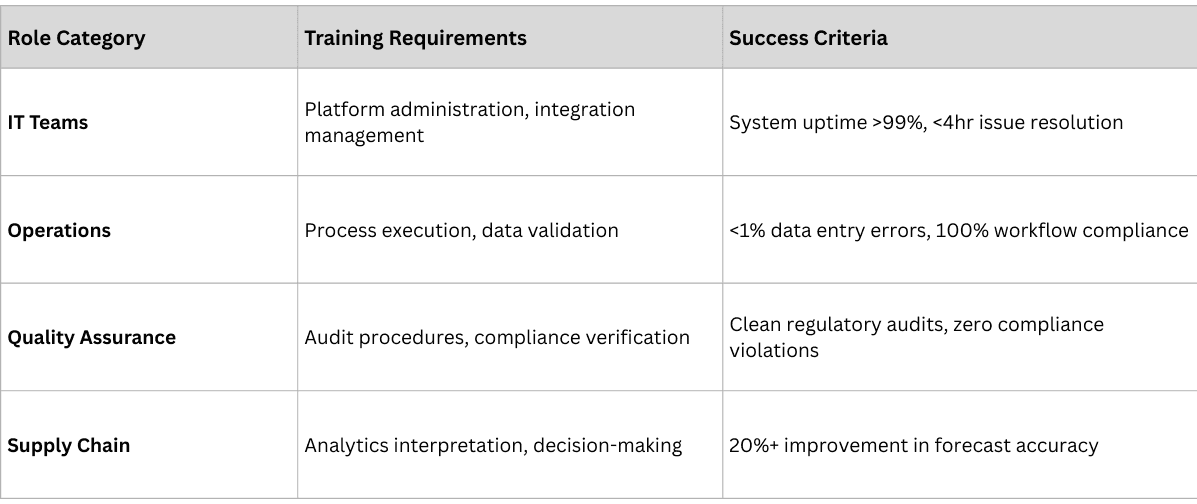
External Partner Onboarding:
- Distributor Enablement: Technology deployment, process training, performance monitoring
- Pharmacy Integration: System connectivity, workflow adaptation, support provision
- Logistics Coordination: Real-time data sharing, exception handling, performance tracking
Change Management Strategy
Successful adoption requires comprehensive change management addressing both technical and cultural aspects:
Communication Framework:
- Executive Sponsorship: Visible leadership commitment and strategic messaging
- Regular Updates: Bi-weekly progress communications and success story sharing
- Feedback Mechanisms: Structured channels for user input and continuous improvement
- Recognition Programs: Celebrating early adopters and implementation milestones
Training and Support:
- Role-Specific Training: Customized programs for different user types and responsibilities
- Hands-On Practice: Simulated environments for safe learning and skill development
- Ongoing Support: Help desk, documentation, and peer support networks
- Continuous Learning: Regular updates on new features and best practices
Phase 4: Pilot Implementation (Months 5-8)
The pilot phase validates the solution with a limited scope before full-scale deployment.
Pilot Scope Selection Criteria:
- Representative Products: Include high-value, high-volume, and complex products
- Diverse Geography: Test in different regulatory environments and market conditions
- Varied Stakeholders: Include different distributor types, pharmacy formats, and customer segments
- Manageable Scale: Large enough to validate capabilities, small enough to manage risks
Pilot Success Framework
Risk Mitigation Strategies:
- Parallel Operations: Run new and old systems simultaneously during transitionm
- Rollback Procedures: Detailed plans for reverting to previous state if needed
- Escalation Protocols: Clear procedures for rapid issue resolution
- Stakeholder Communication: Proactive updates on pilot progress and lessons learned
Pilot Evaluation and Optimization:
The pilot phase should include structured evaluation and optimization cycles:
Week 2 Checkpoint:
- Initial user feedback collection
- Technical performance validation
- Quick fixes and configuration adjustments
Month 1 Review:
- Comprehensive performance analysis
- User adoption assessment
- Process refinement implementation
Month 2 Optimization:
- Advanced feature activation
- Integration enhancement
- Scaling preparation
Phase 5: Full-Scale Deployment (Months 7-12)
The deployment phase scales the solution across the entire organization and supply chain.
Deployment Strategy Options:
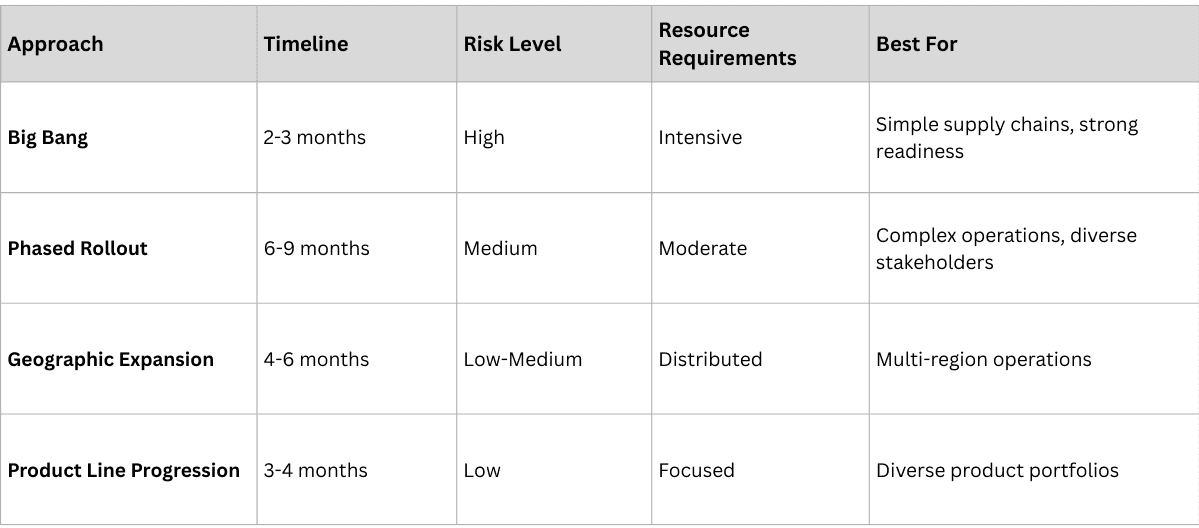
Deployment Execution Framework:
Pre-Deployment Preparation:
- Final system testing and performance validation
- Stakeholder readiness confirmation and sign-off
- Support team preparation and resource allocation
- Communication campaign launch and awareness building
Deployment Execution:
- Coordinated go-live activities across all stakeholders
- Real-time monitoring and issue response
- User support and immediate problem resolution
- Performance tracking and optimization
Post-Deployment Stabilization:
- 30-day performance monitoring and optimization
- User feedback collection and system refinement
- Advanced feature activation and capability expansion
- Success measurement and ROI validation
Critical Success Factors
Our experience across multiple implementations has identified several factors that consistently determine success:
Executive Sponsorship and Governance
Strong Leadership Commitment:
- Successful implementations require visible, consistent executive sponsorship that:
- Communicates strategic importance and expected benefits
- Resolves cross-functional conflicts and resource constraints
- Maintains momentum through inevitable challenges and setbacks
- Celebrates milestones and recognizes contributor efforts
Effective Governance Structure:
- Steering Committee: Senior executives providing strategic oversight and decision-making authority
- Project Management Office: Day-to-day coordination, timeline management, and resource allocation
- Technical Working Groups: Subject matter experts addressing specific implementation challenges
- User Advisory Groups: Representative stakeholders providing feedback and guidance
Technology Platform Considerations
Cloud-Native Architecture:
Modern traceability implementations should leverage cloud platforms for:
- Scalability: Ability to handle varying transaction volumes and geographic expansion
- Reliability: Enterprise-grade uptime and disaster recovery capabilities
- Security: Advanced security controls and compliance certifications
- Cost Efficiency: Reduced infrastructure investment and maintenance overhead
GS1 Standards Compliance:
Ensure full compliance with relevant GS1 standards:
- GTIN (Global Trade Item Number): Unique product identification
- SSCC (Serial Shipping Container Code): Logistics unit identification
- GLN (Global Location Number): Location and entity identification
- EPCIS (Electronic Product Code Information Services): Event data sharing
Integration Capabilities:
The platform must integrate seamlessly with existing systems:
- Real-Time Data Exchange: Immediate synchronization with ERP, MES, and CRM systems
- Standardized APIs: Enable future integrations and ecosystem expansion
- Data Quality Controls: Automated validation and error correction capabilities
- Audit Trail Maintenance: Complete traceability of all system interactions
Zelthy's traceability and supply chain platform was built with these implementation principles in mind, layering AI intelligence on top of regulatory-grade serialisation infrastructure to deliver both compliance and operational returns.
Data Management and Quality
Data Governance Framework:
- Data Standards: Consistent formats, definitions, and quality criteria
- Data Ownership: Clear accountability for data accuracy and maintenance
- Data Security: Access controls, encryption, and privacy protection
- Data Lifecycle: Retention policies, archival procedures, and deletion protocols
Quality Assurance Processes:
- Automated Validation: Real-time data quality checks and error flagging
- Exception Handling: Procedures for managing data discrepancies and corrections
- Performance Monitoring: Continuous tracking of data quality metrics
- Continuous Improvement: Regular review and enhancement of data processes
Implementation Budget Planning
Understanding the true cost of traceability implementation is crucial for realistic planning and stakeholder expectations.
Cost Structure Analysis
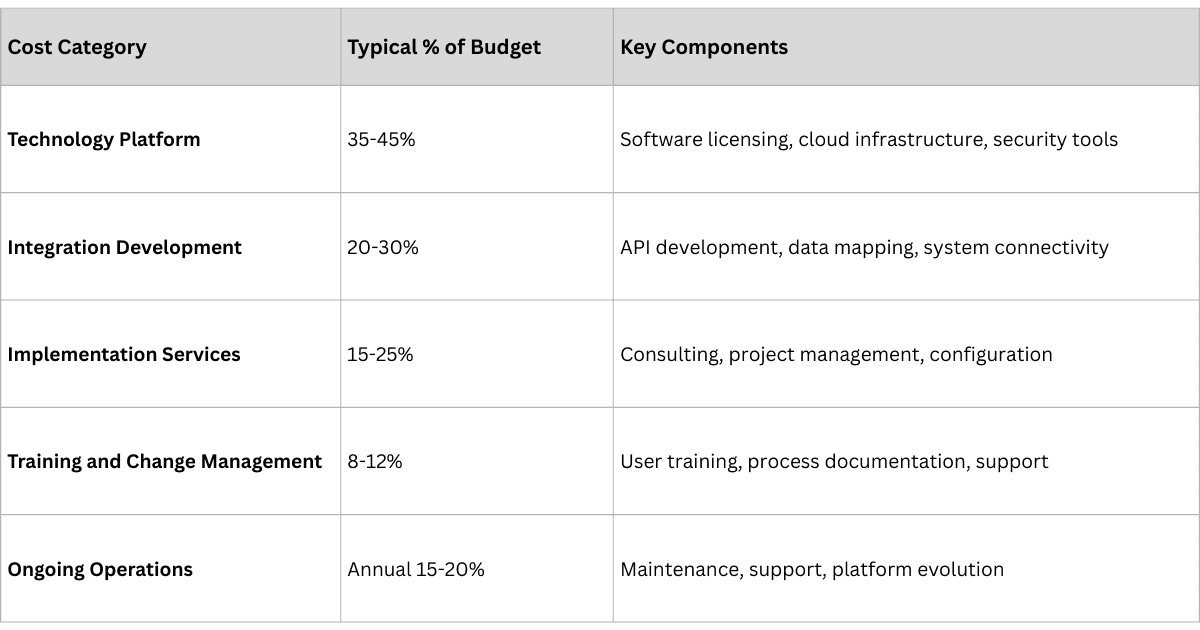
Implementation costs vary by company size:
- SMEs ($100M–$1B revenue) typically invest $500K–$2M over 6–12 months
- large enterprises ($1B–$10B) invest $2M–$8M over 12–18 months
- multinationals ($10B+) invest $8M–$25M over 18–24 months.
Key cost categories include technology platform licensing, system integration, stakeholder training, and ongoing operations and support.
Budget Planning Framework
Small-Medium Enterprise (Annual Revenue $100M-$1B):
- Total Implementation Cost: $500K - $2M
- Timeline: 6-12 months
- Focus: Compliance achievement with operational efficiency gains
Large Enterprise (Annual Revenue $1B-$10B):
- Total Implementation Cost: $2M - $8M
- Timeline: 12-18 months
- Focus: Advanced analytics and competitive advantage creation
Multinational Corporation (Annual Revenue $10B+):
- Total Implementation Cost: $8M - $25M
- Timeline: 18-24 months
- Focus: Global platform with regional customization and innovation
ROI Expectations and Timeline
Immediate Benefits (Months 1-6):
- Regulatory compliance achievement
- Reduced manual processes and associated errors
- Improved audit readiness and inspection outcomes
Short-Term Benefits (Months 6-18):
- 15-25% improvement in inventory management
- 10-20% reduction in product recalls and investigations
- 20-30% faster regulatory reporting and compliance activities
Long-Term Benefits (Months 18+):
- 25-40% improvement in demand forecasting accuracy
- 10-15% reduction in working capital requirements
- New revenue opportunities through data-driven services
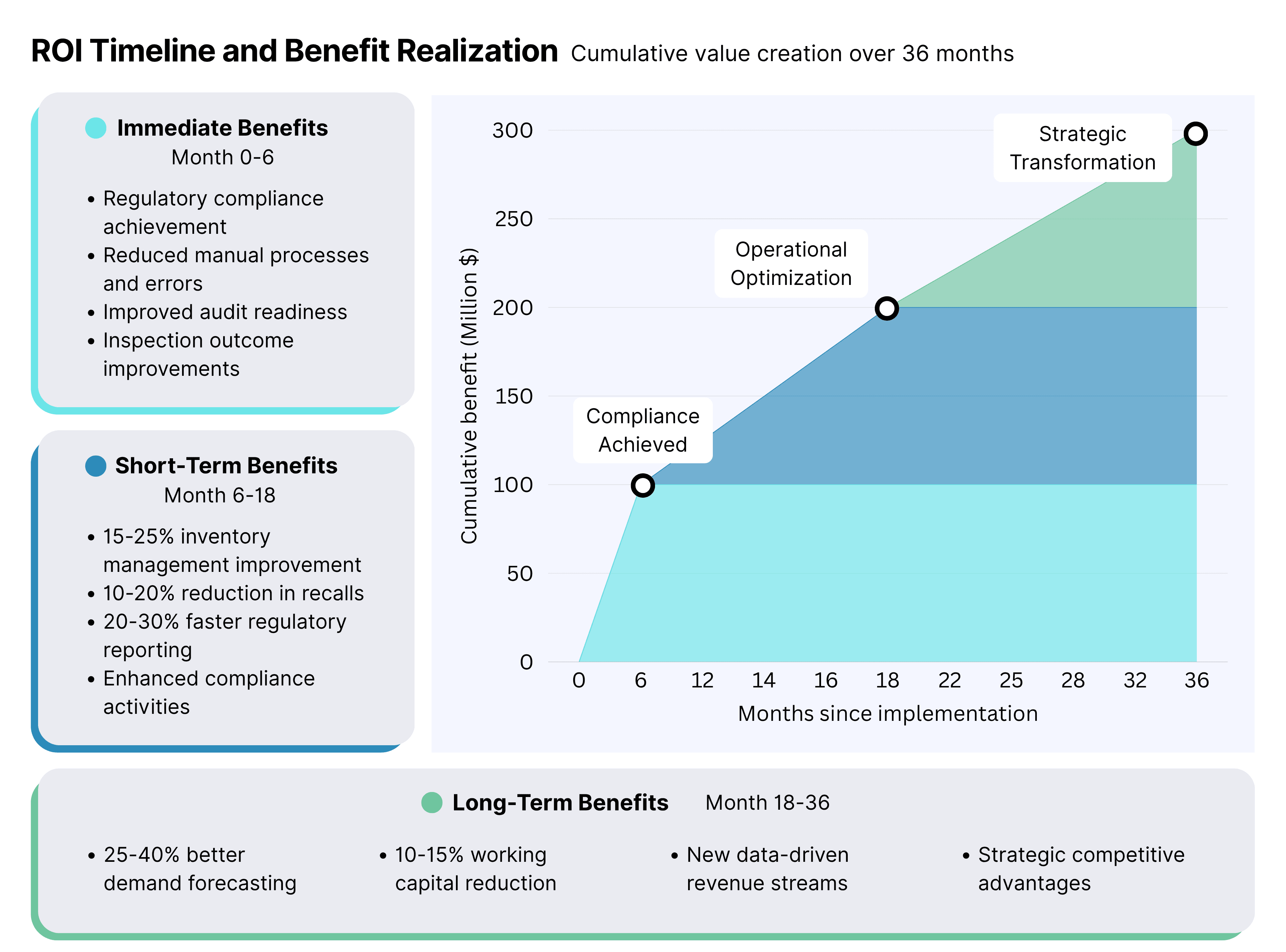
- Immediate benefits (months 1–6) include regulatory compliance and audit readiness.
- Short-term benefits (months 6–18) include 15–25% inventory improvement, 10–20% fewer recalls, and 20–30% faster regulatory reporting.
- Long-term benefits (months 18+) include 25–40% better demand forecasting, 10–15% working capital reduction, and new data-driven revenue opportunities.
Common Implementation challenges and Solutions
Even well-planned implementations face predictable challenges. Understanding these in advance enables proactive mitigation strategies.
Challenge 1: Data Quality and Standardization
Problem
Inconsistent data formats, incomplete information, and quality issues across different systems and stakeholders.
Solution Framework:
- Data Mapping and Cleansing: Comprehensive assessment and standardization of existing data
- Validation Rules: Automated checks for completeness, accuracy, and format compliance
- Training Programs: User education on data entry standards and quality requirements
- Continuous Monitoring: Ongoing performance tracking and quality improvement processes
Challenge 2: Stakeholder Resistance and Adoption
Problem
Users resistant to change, inadequate training, and poor adoption of new processes.
Solution Framework:
- Early Engagement: Involve stakeholders in requirements gathering and solution design
- Value Communication: Clear articulation of benefits and impact on daily work
- Comprehensive Training: Role-specific programs with hands-on practice and ongoing support
- Incentive Alignment: Recognition programs and performance metrics that encourage adoption
Challenge 3: Integration Complexity
Problem
Difficulty connecting with legacy systems, data format incompatibilities, and performance issues.
Solution Framework:
- Phased Integration: Gradual connection of systems with thorough testing at each stage
- Middleware Solutions: Use of integration platforms to manage complexity and reduce risk
- API Strategy: Standardized interfaces for future connectivity and ecosystem expansion
- Performance Optimization: Load testing and optimization to ensure acceptable response times
Challenge 4: Regulatory Compliance Complexity
Problem
Varying requirements across different markets, evolving regulations, and audit readiness.
Solution Framework:
- Regulatory Expertise: Engagement of specialists familiar with relevant requirements
- Compliance Mapping: Detailed analysis of requirements and solution capabilities
- Audit Preparation: Structured processes for maintaining compliance evidence
- Regulatory Monitoring: Ongoing tracking of requirement changes and impact assessment
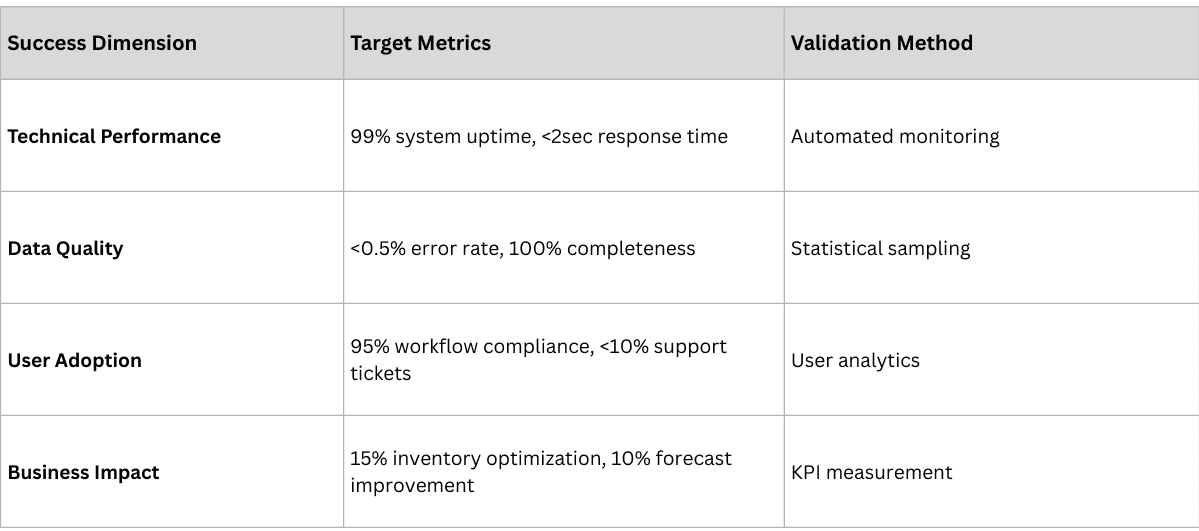
Measuring Implementation Success
Successful implementations require comprehensive measurement frameworks that track both project execution and business impact.
Project Execution Metrics
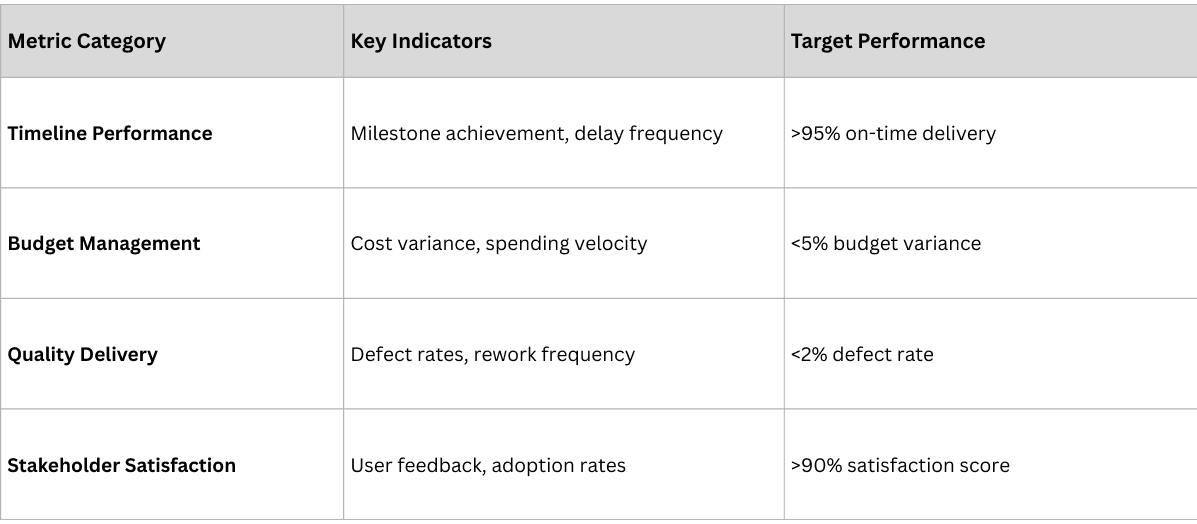
Business Impact Metrics
Operational Excellence:
- Data quality improvement (target: >98% accuracy)
- Process efficiency gains (target: 20-30% reduction in manual effort)
- Error reduction (target: 50-75% fewer compliance violations)
- Audit readiness (target: <48 hours for regulatory responses)
Financial Performance:
- Working capital optimization (target: 10-20% reduction)
- Inventory management improvement (target: 15-25% better turns)
- Cost reduction (target: 5-10% operational cost savings)
- Revenue enhancement (target: new service opportunities)
Strategic Capabilities:
- Market intelligence quality (target: real-time visibility)
- Decision-making speed (target: 50% faster strategic responses)
- Competitive advantage creation (target: measurable market differentiation)
- Innovation enablement (target: new business model opportunities)
For a production example, see how Zelthy's platform delivered 100% chain-of-custody visibility and 15% fewer stockouts for a top-10 pharma company across multiple manufacturing sites and distribution tiers.
Future-Proofing Your Implementation
Successful traceability implementations must be designed for evolution and adaptation as technology, regulations, and business requirements change.
Technology Evolution Considerations
Emerging Technology Integration:
- Artificial Intelligence: Predictive analytics, anomaly detection, automated decision-making
- Blockchain: Enhanced trust, immutable audit trails, smart contract automation
- IoT Integration: Real-time environmental monitoring, automated data capture
- Advanced Analytics: Machine learning, pattern recognition, optimization algorithms
Platform Evolution Strategy:
- Modular Architecture: Enable incremental capability addition without system disruption
- API-First Design: Support future integrations and ecosystem expansion
- Cloud-Native Approach: Leverage continuous platform improvements and scaling
- Standards Compliance: Ensure compatibility with evolving industry standards
Regulatory Adaptability
Global Expansion Readiness:
- Platform flexibility to accommodate different regulatory requirements
- Multi-language and multi-currency support capabilities
- Local compliance management and reporting features
- Regional customization without core system modification
Regulation Evolution Management:
- Monitoring systems for tracking regulatory changes
- Impact assessment processes for requirement modifications
- Agile modification capabilities for rapid compliance adaptation
- Stakeholder communication systems for change management
Conclusion: The Path To Implementation Success
Implementing complete pharmaceutical traceability is neither simple nor straightforward, but it is absolutely achievable with the right approach, preparation, and execution. The companies that succeed follow proven frameworks, learn from others' experiences, and treat implementation as organizational transformation rather than technology deployment.
Key Success Principles
- Strategic Clarity: Define success beyond compliance and align stakeholders around common objectives
- Comprehensive Planning: Address technology, people, processes, and organizational change simultaneously
- Stakeholder Engagement: Involve all participants in design, implementation, and optimization
- Phased Execution: Validate approaches through pilots before full-scale deployment
- Continuous Improvement: Monitor performance and optimize operations based on results
The pharmaceutical industry stands at a critical juncture where traceability implementation is shifting from regulatory requirement to competitive necessity. Companies that implement strategically, execute professionally, and optimize continuously will create lasting advantages that extend far beyond compliance.
The framework, strategies, and lessons shared in this guide represent collective learning from multiple successful implementations. While every implementation has unique characteristics, the fundamental principles of success remain consistent: clear strategy, comprehensive planning, stakeholder alignment, and disciplined execution.
The investment in complete traceability implementation is significant, but the benefits—regulatory compliance, operational excellence, and competitive advantage—make it one of the most important strategic initiatives pharmaceutical companies can undertake today.
Next in the series: How Traceability Transforms Pharma Business Models — why leading companies are treating complete traceability as a competitive weapon, not just a compliance cost.
About This Series
This implementation guide draws from real-world experience deploying traceability solutions across pharmaceutical companies ranging from mid-sized enterprises to global multinationals. The frameworks and strategies presented have been validated through successful implementations serving millions of patients and processing billions of pharmaceutical transactions.
Previous in This Series:
Upcoming:
- Blog 3: How Traceability Transforms Pharma Business Models
- White Paper: Beyond Track and Trace - Strategic Business Model Transformation
For specific questions about traceability requirements in your market or to discuss implementation strategies, feel free to reach out to our team at connect@zelthy.com or directly message us on LinkedIn.
Frequently Asked Questions
Why do most pharmaceutical traceability implementations fail?
Most implementations fail because companies treat traceability as a technology project rather than an organizational transformation. Common failure patterns include technology-first deployment without process redesign, stakeholder misalignment between manufacturers, distributors, and pharmacies, and a compliance-only mindset that builds minimum viable systems. One 11-site study found serialization capital costs averaged €600,000 per line — four times regulatory estimates — yet 88% of sites reported no net productivity gain, reflecting implementation without strategic intent.
What are the five phases of a successful traceability implementation?
A proven five-phase framework structures successful implementations: Phase 1 (Months 1–2) establishes strategic foundations and stakeholder alignment; Phase 2 (Months 2–4) covers platform architecture and integration design; Phase 3 (Months 3–6) focuses on change management and stakeholder enablement; Phase 4 (Months 5–8) runs a controlled pilot across representative products and geographies; Phase 5 (ongoing) drives continuous optimization and value expansion beyond the initial compliance goal.
What does a traceability business case need to include?
A traceability business case must quantify value beyond compliance to secure executive buy-in. It should project operational benefits (demand forecasting accuracy, inventory reduction, stockout prevention), commercial benefits (marketing ROI improvement, market share gains), and risk mitigation benefits (recall cost reduction, supply chain diversion losses prevented). Projects justified solely on compliance cost avoidance consistently underperform in adoption and value realization compared to implementations framed as strategic transformation initiatives.
Why did 52% of pharma plants skip aggregation in DSCSA implementation?
A 2021 PMMI survey found that 52% of pharmaceutical plants implementing US DSCSA standards skipped the aggregation data layer — which maps individual serialized units to cases and pallets — because aggregation was not explicitly mandated. This decision was operationally short-sighted: aggregation is the data layer that enables supply chain analytics, demand sensing, and diversion detection. Plants that skipped aggregation achieved regulatory compliance but forfeited the primary operational intelligence benefits that justify traceability investment.
How should pharmaceutical companies select a traceability platform?
Platform selection should follow architecture-first principles: cloud-native scalability, API-enabled integration with existing ERP, MES, WMS, and analytics systems, GS1 data standards compliance for interoperability, and security by design. Selection criteria should also assess the vendor's ability to support business model use cases — demand sensing, secondary sales analytics, patient engagement integration — not just regulatory reporting. The cheapest compliance platform often becomes the most expensive choice when value expansion is blocked by architectural limitations.
What change management approaches work in traceability rollouts?
Successful traceability change management requires visible executive sponsorship, role-specific training (not generic system training), hands-on practice in simulated environments, and structured feedback channels for frontline users. Bi-weekly progress communications and recognition programs for early adopters accelerate adoption. External partner onboarding — distributors, pharmacies, and logistics providers — requires dedicated enablement tracks, not just API documentation. Bain research indicates only 25% of companies rate their cross-ecosystem collaboration as highly effective, making partner enablement the most underfunded component of traceability programs.
How long does a full pharmaceutical traceability implementation take?
A well-structured end-to-end traceability implementation — from strategic foundation through pilot validation — typically spans 8–9 months, with continuous optimization extending beyond initial go-live. Strategic foundation and architecture design run in parallel across months 1–4, stakeholder enablement overlaps from months 3–6, and the pilot phase runs months 5–8. Organizations implementing complex multi-jurisdictional programs with ERP and MES integration should budget 12–18 months for the pilot phase alone, particularly when external partner onboarding involves multiple distributor systems.
References and Sources
- R.G. Mukunda, N. Vishal Gupta. “Pharma Serialization: Implementation and Challenges.” International Journal of Pharmaceutical Sciences Review and Research, 2015.
- McKinsey & Company. "Digital transformation: Health systems’ investment priorities." Healthcare Industry Report, 2024.
- McKinsey & Company. “Rewired pharma companies will win in the digital age.” Healthcare Industry Report, 2023.
- GS1 US. “Applying GS1 System of Standards for DSCSA and Serialized Interoperable Traceability | Implementation Guideline.” 2022.
- GS1 US. “Implementation Guideline: Applying GS1 Standards for DSCSA and Traceability.” 2016.
- U.S. Food and Drug Administration. "FDA’s Implementation of Drug Supply Chain Security Act (DSCSA) Requirements.” 2024.
- The European Parliament and the Council of the European Union. "Falsified Medicines Directive." 2011.
- European Commission. “Implementation measures by the commission in context of the falsified medicines directive.”
- GS1 Healthcare. "Healthcare Supply Chain Traceability.” Industry Report.
- Accenture. "Achieving high performance in pharmaceuticals through supply chain analytics."
- Deloitte Insights. Deloitte Centre for Health solutions. "Intelligent drug supply chain: Creating value from AI." 2024.
- Deloitte Insights. Deloitte Centre for Health Solutions. “Pharma’s supply chain workforce.” 2024.
- Vergara, I. T. & McKinnell, D. “Optimising pharmaceutical supply chain digitalisation through serialisation and traceability: A perspective beyond compliance.” Deloitte Blogs, 2023.
- TraceLink. “Global Drug Supply, Safety and Traceability Report.” Industry Report, 2025.
- ISPE (International Society for Pharmaceutical Engineering). "Good Practice Guide: Digital Validation." Technical Guidelines, 2025.
- ISPE (International Society for Pharmaceutical Engineering). "GAMP 5: A Risk-Based Approach to Compliant GxP Computerized Systems." 2022.
- Alphena Pharma Solutions. “Track and Trace: A Step-by-Step Guide to Implementing an Effective Program.”
- Boston Consulting Group. "Building the Supply Chain of the Future." Strategy Report, 2023.
- O' Mahony, D., Lynch, A., & McDermott, O. (2024). The Impact of Serialisation on Operational Efficiency and Productivity in Irish Pharmaceutical Sites. Therapeutic innovation & regulatory science, 58(5), 883–896. Available at: https://doi.org/10.1007/s43441-024-00662-1
- Hernan Saenz, Joshua Hinkel, and Tessa Bysong. “Traceability: The Next Supply Chain Revolution.” Bain & Company. 2021.
- “Serialization will impact all drug manufacturing.” Healthcare Packaging, 2017.